|
To obtain a +1 charge it loses an electron, resulting in a configuration of Ar. The electron configuration of potassium is 1s2 2s2 2p6. One important aspect of potassium is its electron configuration, which determines its chemical behavior. It belongs to the alkali metal group and is highly reactive, readily forming compounds with various elements. Their electron configurations are 1 s 1 and 1 s 2, respectively with He, the n 1 shell is filled. Kung ikaw ay mahilig magsugal, ,marahil alam mo na rin ang mga lumang pamahiin na pinaniniwalaan tungkol dito. Both of these alloys produce metals of low vapor pressure and melting points. Potassium (K) is orignially in the electron configuration of Ar4s1. Potassium is a chemical element with the symbol K and atomic number 19. The shape of the periodic table mimics the filling of the subshells with electrons. electron configuration for potassium : 3585. /potassiumatom-58b602485f9b5860464c55ea.jpg)
The elements in group-18 of the periodic table are inert gases. 
Therefore, the valence electrons of krypton are eight. This electron configuration shows that the last shell of krypton has eight electrons. Because the reaction attains equilibrium quickly, potassium can be removed continuously as a product in order to shift equilibrium to the right and produce even more potassium in its place.Īlloys of potassium include \(NaK\) (Sodium) and \(KLi\) (Lithium). Ground state electron configuration of krypton is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 6. The reaction is heated in a special device equipped with a furnace, heat-exchanger tubes, a fractionating column, a \(KCl\) feed, a waste removal system, and a vapor condensing system. The Aufbau principle states that an electron occupies orbitals in order from lowest energy to highest.\] When we write the configuration we'll put all 19 electrons in orbitals around the nucleus of the Potassium atom. As we proceed with atoms with multiple electrons, those electrons are added to the next lowest sublevel: \(2s\), \(2p\), \(3s\), and so on. In order to write the Potassium electron configuration we first need to know the number of electrons for the K atom (there are 19 electrons). The single electron of the hydrogen atom will occupy the \(1s\) orbital when the atom is in its ground state. One atom will have 2 electrons in its 1st shell, 8 electrons in its 2nd shell, 8. 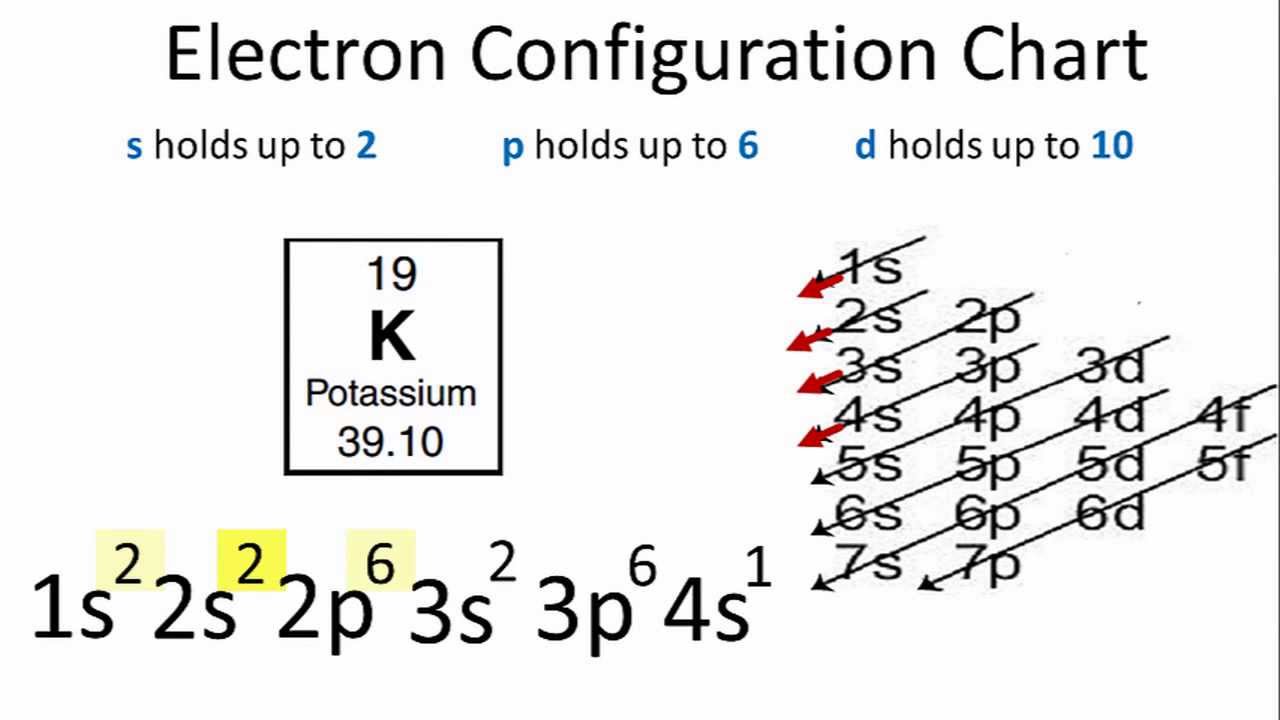
These electron configurations make helium and neon very stable. Similarly, neon has a complete outer 2n shell containing eight electrons. In this table, you can see that helium has a full valence shell, with two electrons in its first and only, 1n, shell. Potassium Potassium X-ray photoelectron spectra, potassium electron configuration, and other elemental information. In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. Examples of some neutral atoms and their electron configurations are shown below. The lowest energy sublevel is always the \(1s\) sublevel, which consists of one orbital. Neutral potassium atoms also have 19 electrons surrounding their nuclei. Electron atomic and molecular orbitals A Bohr diagram of lithium. \) shows the order of increasing energy of the sublevels.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
